Regulatory Perspectives on Precision Dosing & Cardiac Safety: Insights from Dr. Robert Temple (FDA)
Archival Context: This document provides a comprehensive summary and scientific expansion of the presentation delivered by Dr. Robert Temple, Deputy Center Director for Clinical Science at the FDA’s Center for Drug Evaluation and Research (CDER). The presentation, given at the Cardiac Safety Research Consortium (CSRC) Annual Meeting on December 3, 2015, addressed the critical intersection of precision medicine, anticoagulant dosing strategies, and real-world safety evidence.
Executive Summary
The traditional paradigm of drug development has long adhered to a “one-size-fits-all” dosing philosophy. However, as pharmacological science advances, it has become increasingly evident that fixed-dose regimens often fail to account for the complex physiological variability inherent in real-world patient populations. This is particularly true for drugs with a narrow therapeutic index, such as the Direct Oral Anticoagulants (DOACs).
In his seminal 2015 address, Dr. Robert Temple highlighted the regulatory challenges associated with Rivaroxaban and other Factor Xa inhibitors. While randomized clinical trials (RCTs) establish efficacy, they often exclude patients with significant comorbidities, renal impairment, or concomitant medication use.
This article reviews the regulatory push towards precision dosing strategies and explores the pre-clinical tools—including pharmacokinetic (PK) modeling, metabolic stability assays, and high-purity reference inhibitors—that are essential for validating these strategies. Understanding the mechanistic basis of drug clearance and interaction is not only a regulatory requirement but a fundamental step in ensuring cardiac and systemic safety.
Part I: The Regulatory Shift – From Population Averages to Individualized Safety
Dr. Temple’s discussion at the CSRC meeting marked a pivotal moment in regulatory thinking. Historically, the FDA approved drugs based on population averages derived from Phase III trials. If the average patient showed a benefit with acceptable safety, the drug was approved at a standard dose.
The Limitation of Fixed Dosing
For many therapeutic classes, such as analgesics or antihistamines, a wide therapeutic window allows for broad dosing. However, for cardiovascular agents, particularly anticoagulants, the margin for error is slim.
- Under-dosing leads to thrombotic events (stroke, pulmonary embolism).
- Over-dosing leads to life-threatening hemorrhagic events.
Dr. Temple pointed out that “real-world” patients often differ significantly from “trial” patients. Factors such as age, weight, renal function (creatinine clearance), and genetic polymorphisms in metabolic enzymes (e.g., CYP2C9, CYP3A4) create massive variability in drug exposure.
Precision Dosing as a Safety Mandate
The concept of “Precision Dosing” championed by the FDA involves using patient-specific characteristics to tailor the dosage. This approach relies heavily on Exposure-Response (E-R) analysis. By modeling how drug concentration correlates with both efficacy and toxicity, regulators can recommend dose adjustments for specific subpopulations (e.g., reducing the dose in patients with renal failure).
Research Implication: To build robust E-R models, pharmaceutical developers must generate precise pre-clinical data. This necessitates the use of small molecule inhibitors to map metabolic pathways and identify potential drug-drug interactions (DDIs) long before the drug enters human trials.
Part II: Rivaroxaban and the Factor Xa Mechanism
A significant portion of the 2015 discussion focused on Rivaroxaban, a pioneering Factor Xa inhibitor. Understanding why precision dosing is crucial for this drug requires a deep dive into its mechanism of action and chemical properties.
Mechanism of Action
Rivaroxaban works by selectively and reversibly inhibiting Factor Xa, the driver of thrombin generation in the coagulation cascade. Unlike warfarin, which inhibits the synthesis of Vitamin K-dependent clotting factors, Rivaroxaban directly targets the active site of Factor Xa.
[INSERT IMAGE HERE: Chemical Structure of Rivaroxaban]
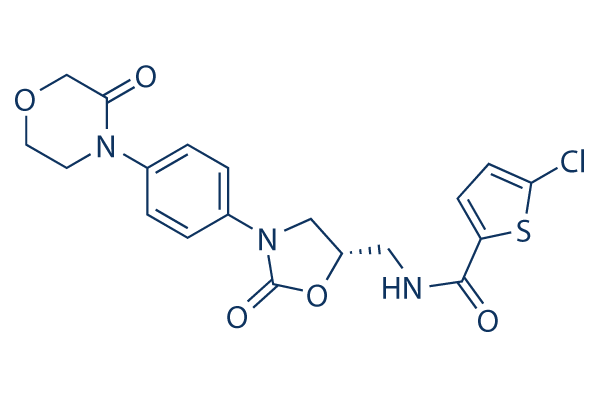
- Caption: Figure 1. The molecular structure of Rivaroxaban. Researchers utilize high-purity chemical standards of this compound to validate bioanalytical methods and conduct metabolic stability screenings.
Why “One Dose” Was Challenging
While Rivaroxaban was marketed with the convenience of fixed dosing (no INR monitoring required), Dr. Temple noted that plasma concentrations can vary up to 50% among individuals. This variability is driven by:
- Absorption: Dependent on P-glycoprotein (P-gp) transporters.
- Metabolism: Primarily metabolized by the cytochrome P450 enzymes (CYP3A4/5 and CYP2J2).
- Elimination: Approximately one-third is excreted unchanged in the urine.
This complex disposition profile means that a patient taking a strong P-gp inhibitor (like ketoconazole) or a CYP3A4 inhibitor (like ritonavir) could experience dangerously high levels of Rivaroxaban, leading to bleeding.
Part III: Pharmacokinetics (PK) and Drug-Drug Interactions (DDIs)
The cornerstone of the precision dosing strategy discussed in Clinical and Translational Science is the accurate prediction of Drug-Drug Interactions (DDIs).
The Role of CYP Enzymes and Transporters
Regulatory guidelines now mandate rigorous screening of new chemical entities (NCEs) against a panel of metabolizing enzymes and transporters. Dr. Temple emphasized that understanding mechanistic interactions is key to writing safe labels.
Modeling Real-World Scenarios
Using Physiologically Based Pharmacokinetic (PBPK) modeling, researchers can simulate “virtual trials.” For example, what happens if a patient taking Rivaroxaban also starts taking Erythromycin (a moderate CYP3A4 inhibitor)? To validate these simulations, laboratory scientists perform in vitro inhibition assays. They incubate the drug with human liver microsomes in the presence of specific inhibitors to measure the change in intrinsic clearance.
Part IV: Pre-Clinical Validation Tools – The Bridge to Safety
Note: This section connects the regulatory concepts to the laboratory tools (and Selleckchem’s product lines), explaining how the data Dr. Temple relies on is actually generated.
The “Precision Dosing” strategy advocated by the FDA is only as good as the data fed into the models. This data generation begins in the discovery lab, utilizing high-throughput screening and specific chemical probes.
1. Metabolic Stability Assays
Before a Factor Xa inhibitor reaches the clinic, its half-life and clearance must be established.
- Methodology: Compounds are incubated with hepatocytes or liver microsomes.
- The Tool: High-purity reference compounds are essential here. Researchers use isotope-labeled standards or pure Rivaroxaban to calibrate Liquid Chromatography-Mass Spectrometry (LC-MS) instruments, ensuring precise quantification of the parent drug and its metabolites.
2. Coagulation Cascade Screening
To ensure safety, a drug must be selective. It should inhibit Factor Xa but spare other proteases like Thrombin (Factor IIa) or Trypsin to avoid off-target toxicity.
- The Tool: Compound Libraries. Researchers utilize protease inhibitor libraries to screen the drug candidate against a wide panel of related enzymes. This “selectivity profiling” is a critical component of the Investigational New Drug (IND) application submitted to the FDA.
[INSERT IMAGE HERE: Coagulation Cascade Diagram]
Figure 2. The Coagulation Cascade. Precision dosing relies on understanding the specific inhibition of Factor Xa. Researchers use selective inhibitors and chromogenic assays to quantify anticoagulant activity in vitro.
3. Transporter Interaction Studies
As noted in the Rivaroxaban case study, transporters like P-gp play a gatekeeper role.
- Methodology: Transporter inhibition assays are conducted using cell lines overexpressing specific transporters (e.g., MDCK-MDR1 cells).
- The Tool: Specific small molecule inhibitors (such as Cyclosporin A or Quinidine) are used as positive controls to verify that the assay system is functioning correctly. Without these validated controls, the FDA will not accept the data.
Part V: Cardiac Safety Beyond the QT Interval
While the CSRC is historically focused on proarrhythmia (QT prolongation), Dr. Temple’s 2015 presentation broadened the definition of “Cardiac Safety” to include hemodynamic stability and bleeding management.
Bleeding as a Cardiovascular Safety Endpoint
In trials like ROCKET AF (Rivaroxaban), major bleeding is a primary safety endpoint. Precision dosing aims to minimize this risk. Dr. Temple argued that analyzing “exposure-response” for bleeding is just as important as analyzing it for efficacy.
The Future: Biomarker-Guided Dosing?
The presentation hinted at a future where dosing might be guided not just by clinical factors (age/weight) but by biomarkers.
- Anti-Xa Activity: While routine monitoring isn’t required for DOACs, measuring Anti-Xa activity can be useful in emergency situations (e.g., overdose or urgent surgery).
- Genetic Screening: Genotyping patients for CYP2C9 or VKORC1 variants (for Warfarin) is the archetype of precision dosing. Similar approaches may eventually apply to newer agents, requiring diagnostic reagents and chemical probes to validate these genetic associations.
Conclusion
Dr. Robert Temple’s 2015 address remains a cornerstone text for understanding the modern regulatory landscape of anticoagulant therapy. It marked a definitive shift away from empirical, fixed dosing towards a mechanistic, evidence-based approach rooted in precision medicine.
For the pharmaceutical scientist, the takeaways are clear:
- Robust Pre-clinical Data is Mandatory: The FDA expects a detailed map of CYP metabolism and transporter interactions.
- Tools Matter: The generation of this data relies on the availability of high-quality chemical inhibitors, reference standards, and compound libraries.
- Safety is Holistic: Cardiac safety encompasses not just electrophysiology, but the entire pharmacokinetic profile of the drug.
As we move forward, the integration of real-world evidence with rigorous in vitro validation will continue to refine dosing strategies, ultimately ensuring that patients receive the maximum therapeutic benefit with the minimum risk.
References & Further Reading
- Konicki, R. et al. “Rivaroxaban Precision Dosing Strategy for Real‐World Atrial Fibrillation Patients.” Clinical and Translational Science (2020).
- Temple, R. “Regulatory Perspectives on Precision Medicine.” CSRC Annual Meeting Presentation, Washington DC, Dec 3, 2015.
- Gintant, G. et al. “The evolving role of specificity and selectivity assays in cardiac safety pharmacology.” Nature Reviews Drug Discovery (2016).
- FDA Guidance for Industry: In Vitro Metabolism- and Transporter-Mediated Drug-Drug Interaction Studies (2020).
